How Many Orbitals Are There in the 4p Subshell
Therefore you can say that a 4p orbital can hold a maximum of two electrons and the 4p subshell can hold a maximum of six electrons. The s subshell has 1 orbital that can hold up to 2 electrons the p subshell has 3 orbitals that can hold up to 6 electrons the d subshell has 5 orbitals that hold up to 10 electrons and the f.

Quantum Numbers N L M S Describe The Properties Of An Atom S Electron Configuration Each Electron In An Atom Can Be Described Completely By These N Quimica
There are NO p orbitals in Level 1 only s orbitals.

. Remembering that the s subshell as 1 orbital and can hold 2 electrons the p subshell has 3 orbitals and can hold 6 electrons the d subshell has 5 orbitals and can hold up to 10 electrons and the f subshell has 7 orbitals can hold up to 14 electrons. Each orbital can hold two electrons so the. Always remember that the magnetic quantum determines the number of orbitals in the subshells and generally s-orbital contains one orbital only as the value of azimuthal quantum number is zero p-orbital contains three orbitals d-orbital contains five orbitals and f-orbital contains seven orbitals.
How many orbitals are there in 4p subshell. Three possible orientations - There are five possible orbitals in a d subshell and 7 possible orbitals in an f subshell. 9 How many orbitals are present in M shell.
With f subshell l3 so ml -3-2 -1 0 123 so there are seven possible values of ml Remember that 4 is the number of principal quantum number it has nothing to do with the number of the orbitals. For any atom there are seven 5f orbitals. 7 How many orbitals are there in period 4.
There are 2l1 such values of ml for a given l and thus there are 2l1 orbitals in each subshell. Now the 4p subshell contains a total of three 4p orbitals 4px 4py and 4pz. It does not matter if your energy level that is the coefficientnumber before the spdf orbital goes as high as 7 which is by far the maximum the number of suborbitals in p is always three.
Maximum 6 electrons in 3 orbitals. How many orbitals are there in the 4p subshell quizlet. Do not confuse the number of orbitals in a subshell with the number of electrons the subshell can hold.
In the hydrogen atom which of the following orbitals has the lowest energy. What element is 3s2 3p1. There are only ever 3 p orbitals in a level there are 3 p orbitals in the 3p sublevel 4p sublevel etc.
How many orbitals are 5f. So there will seven 4f orbitals. How many orbitals are in a 3d subshell.
4 subshells There are 4 subshells s p d and f. 13 What does 2s orbital mean. The f-orbitals are unusual in that there are two sets of orbitals in common use.
This is the best answer based on feedback and ratings. Hence total number of electrons 7214. What are the implications for a political system if it is left unclear whether elected legislators are responsible for functioning as.
View the full answer. There are 32 electrons in the 4th energy shell so there must be 16 orbitals in the 4th shell as the number of orbitals is half the number of electrons. The 4p subshell has 3.
11 How many orbitals are in the second shell of neon. The 4p subshell contains 4px 4py and 4pz orbitals. How many orbitals are there in 4p sublevel.
How many electrons can be accommodated in the 4p subshell. 18 QUESTIONS What is the electron configuration of fluorine. How many orbitals are there in a 4p subshell.
10 How many orbitals are there in the sixth shell. Therefore the 4p orbital can hold two electrons and the 4p subshell can hold a total of six electrons. Critical Thinking Representation is a critical concept in democratic thinking.
What is true about the atom of cobalt-60. 2 The number of orbitals present in an electron subshell is always six. Thus the answer is B.
The px py and pz each of which needs a maximum of two electrons of opposite spins as per the Aufbau Hunds and Pauli exclusion principles. Therefore we can say that there are a total of three orbitals in 4p subshell. As each orbital can hold maximum two electrons so total electrons p orbital can hold is 6.
What are the N and L quantum numbers for the 4p subshell. Take a look at the illustration. 3 An atom as a whole is electrically neutral because all charged subatomic particles reside in asked Jun 25 2017 in Chemistry by littleone.
Cobalt-60 is a radioactive isotope sometimes used in there warmer of cancer. 2 C3 D4 E5 QUESTION 7 According to the quantum mechanical model how many orbitals in a pven atom have n-3. Therefore the 4p orbital can hold two electrons and the 4p subshell can hold a total.
This number divides the subshell into individual orbitals which hold the electrons. Then total values of m2317 orbitals We know that one orbital contains two electrons. Use shorthand notation NO.
9 OD 10 E. 12 How many orbitals are in the fourth shell. How many orbitals are there in the 4p subshell.
Which of the following subshell contains only one orbital. How many Subshells are in a shell. Each subshell can hold a different number of.
Why 4s orbital is filled before 3d orbital. This tells us that each subshell has double the electrons per orbital. How many orbitals are there in the 4p subshell.
8 How many orbitals are in each subshell. What describes a proton. 14 How many orbitals are in 1s.
There are 2l1 orbitals in each subshell. Explanation with p subshell l 1 ml -1 0 1 so there are three possible values of ml Remember that 4 is the number of principal quantum number it has nothing to do with the number of the orbitals. There is one s orbital and there are three p orbitals five d orbitals and seven f orbitals.
So for part a in a 4p subshell there are 3 orbitals present as the p subshell can hold up to 6 electrons. Thus the s subshell has only one orbital the p subshell has three orbitals and so on. 100 5 ratings 4p subsh.

Electron Subatomic Particle Teaching Chemistry Chemistry Classroom Chemistry Education

Orbitals And Quantum Numbers Practice Questions

Electron Configuration Test Chemistry Quizizz

The Designation Of A Sub Shell With N 4 And L 3 Is Youtube
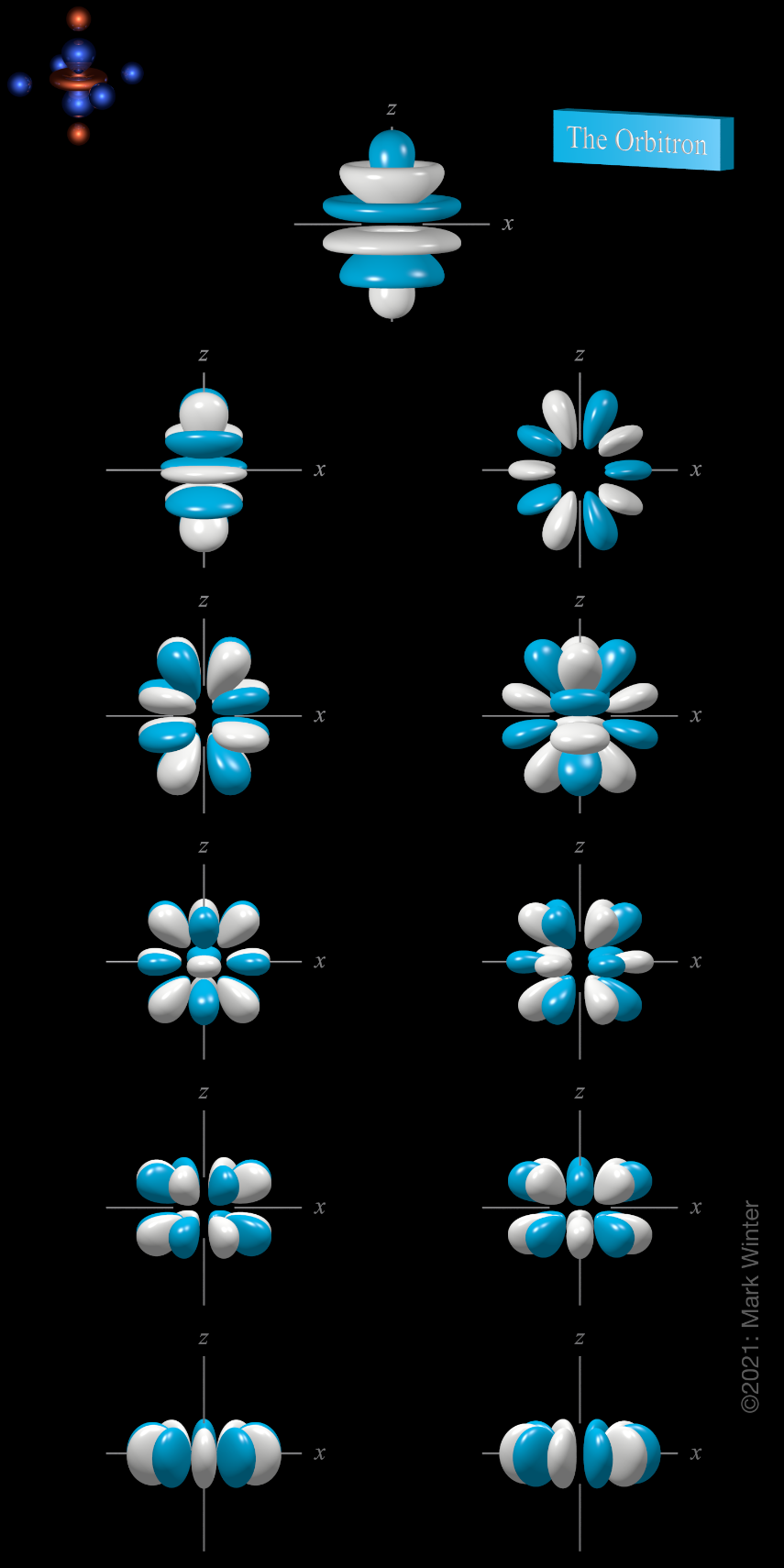
The Orbitron 6h Atomic Orbitals
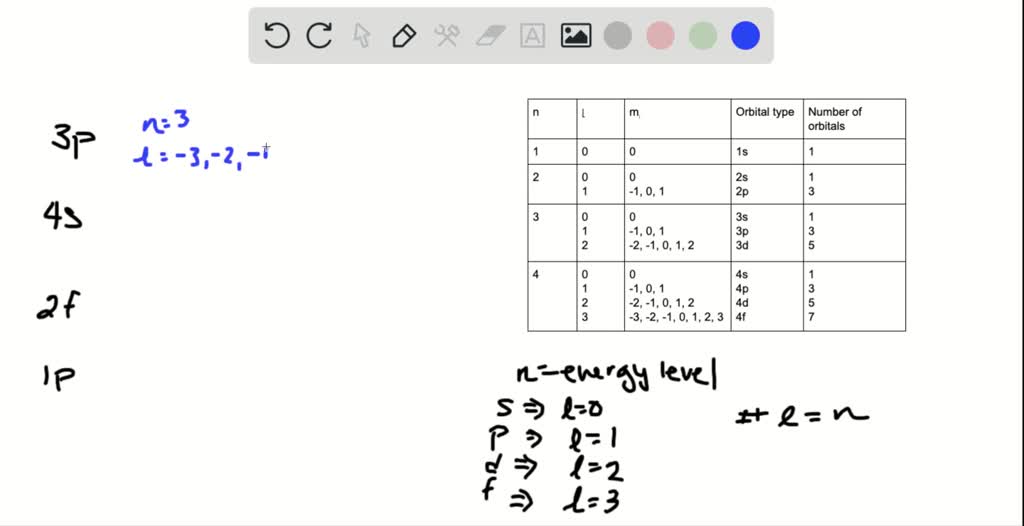
Solved State Which Of The Following Orbitals Cannot Exist According To The Quantum Theory 3 P 4 S 2 F And 1p Briefly Explain Your Answers

Electronic Configurations Electron Configuration Physics And Mathematics Writing Challenge

Electron Configuration How To Memorize Orbitals Sequence Easily For Writting Electron Configuration Y Electron Configuration How To Memorize Things Electrons

How To Write Electron Configurations For Atoms Of Any Element Electron Configuration Electrons Writing

How Many Different Sublevels Are In The Third Energy Level At Level

Orbitals Atomic Energy Levels Sublevels Explained Basic Introduction To Quantum Numbers Youtube

Electron Subshells Orbitals 1 1 7 Cie A Level Chemistry Revision Notes 2022 Save My Exams

Solved A When N 4 Ell 2 And M Ell 1 To What Orbital Type Does This Refer Give The Orbital Label Such As 1 S B How Many Orbitals Occur In The N 5 Electron

How Many Orbitals In 4p Lisbdnet Com

The Electron Configuration Of Elements How Do You Do An Easy Electron Configuration Electron Configuration Aufbau Principle Electrons

How Many Electrons Can The 4th Energy Level Hold At Level

Solved Use Orbital Diagrams To Show The Distribution Of Electrons Among The Orbitals In A The 4 P Subshell Of Br B The 3 D Subshell Of Mathrm Co 2 Given That The Two Electrons

Comments
Post a Comment